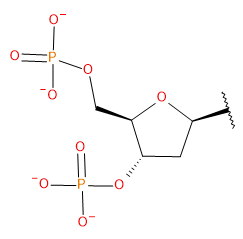
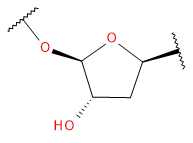
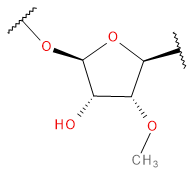
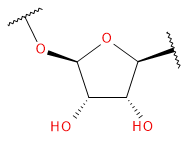
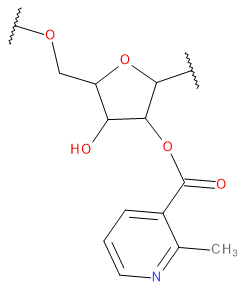
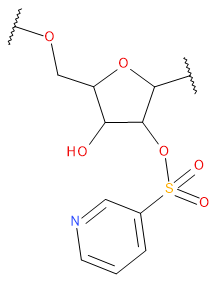
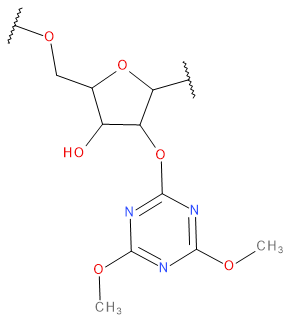
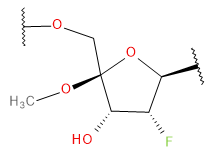
Backbone
Assume that when it is a stereo-isomer, the fragment will have the final number 1 for the (S) configuration and 2 for the (R) configuration.
| FRAGMENT ID | 2D Structure | Name (3D link) | Notes |
| 5PO |  | 5PO | 5′-terminal cap |
| DPO | DPO | Standard dimethyl phosphate (regular backbone) Charge -1 Atom types from OL15/21 | |
| RPO | RPO | Standard dimethyl phosphate (regular backbone) Charge -1 Atom types from OL3 | |
| BPO | 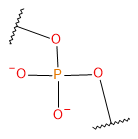 | BPO | |
| PSS | PSS | Phosphorodithioate Charge -1 | |
| PS1 | PS1 (S) | Thiophosphate (S) Charge -1 Other names: phosphorothioate | |
| PS2 | PS2 (R) | Thiophosphate (R) Charge -1 Other names: phosphorothioate | |
| MP1 | MP1 (S) | Methylphosphate (S) | |
| MP2 | MP2 (R) | Methylphosphate (R) | |
| NS1 | NS1 (S) | Thio phosphoramidate (S) Charge -1 | |
| NS2 | NS2 (R) | Thio phosphoramidate (R) Charge -1 | |
| PR1 | PR1 | Propyl backbone (S) | |
| PR2 | PR2 | Propyl backbone (R) | |
| IB1 | IB1 (S) | Isobutyl backbone (S) | |
| IB2 | IB2 (R) | Isobutyl backbone (R) | |
| MO1 | MO1 (S) | MOP backbone (S) | |
| MO2 | MO2 (R) | MOP backbone (R) | |
| PC1 | PC1 (S) | Phosphonoacetate (S) Charge -1 | |
| PC2 | PC2(R) | Phosphonoacetate (R) Charge -1 | |
| MES | 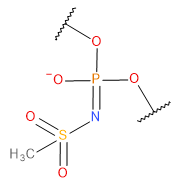 | MES | Charge -1 |
| MXG | MXG | Methylene phosphonate Charge -1 | |
| PN1 | PN1 (S) | Phosphoryl guanidine (S) | |
| PN2 | PN2 (R) | Phosphoryl guanidine (R) | |
| WX1 | MX1 (S) | Mesyl -N-phosphonate (S) Charge -1 | |
| WX2 | MX2 (R) | Mesyl -N-phosphonate (R) Charge -1 | |
| VX1 |  | VX1 (S) | Ethyl-mesyl phosphonate (S) |
| VX2 | 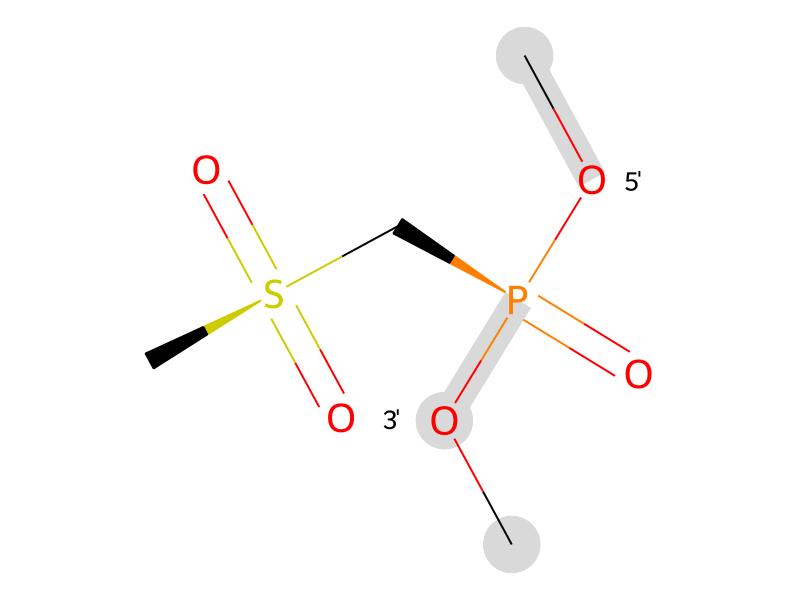 | VX2 (R) | Ethyl-mesyl phosphonate (R) |
| XV1 |  | XV1 (S) | Isopropyl mesyl phosphonate (S) |
| XV2 |  | XV2 (R) | Isopropyl mesyl phosphonate (R) |
| ZV1 |  | ZV1 (S) | Amine-mesyl phosphonate (S) |
| ZV2 |  | ZV2 (R) | Amine-mesyl phosphonate (R) |
Sugar / Linker
A-based modification
C-based modification
| FRAGMENT ID | 2D Structure | Name (3D link) | Notes |
| DCC/RCC | Cytidine (DCC) / Cytidine (RCC) | Ideal module Name HARDCODED Charges from OL3/OL15 | |
| 5FC | 5-fluorocytidine 5′-(dihydrogen phosphate) | ||
| A4C | N4-acetylcytidine | ||
| B5C | 5-bromocytidine 5′-(dihydrogen phosphate) | ||
| BIC | isocytidine-5′-monophosphate | ||
| BPC | agmatidine | ||
| C5C | 5-carboxylcytosine | ||
| E3C | 3-ethylcytosine | ||
| F5C | 5-formylcytosine | ||
| G5C | 5-glucosylmethylcytosine | ||
| H5C | 5-hydroxymethylcytosine | ||
| K2C | 2-lysidine | ||
| M2C | 2-O-methylcytosine | ||
| M3C | 3-methylcytosine | ||
| M4C | N4,N4-dimethylcytidine | ||
| M5C | 5’-methylcytidine | ||
| N3C | N3-protonated cytidine | Charge +1 | |
| N4C | N4-methylcytosine | ||
| N5C | 5-nitrocytidine 5′-(dihydrogen phosphate) | Charge +1 | |
| NEC | 3,N4-ethenocytosine | ||
| O5C | 5-hydroxycytidine | ||
| PPC | 6’-phenylpyrrolo-cytosine | ||
| RYC | 5′-3,6-dihydrocytidylic acid | ||
| S2C | 2-thiocytidine | ||
| FMC | 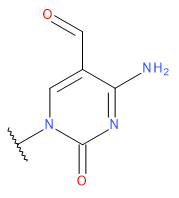 | FMC | |
| FRC | 5-Formyl dC III CE | ||
| FQC | Ac-5-Me-dC-CE | ||
| FSC | 5-Formyl-dC-CE | ||
| FUC | 5-Hydroxymethyl-dC II-CE | ||
| FVC | 5-I-dC-CE | 5′-Dimethoxytrityl-N-benzoyl-5-iodo-2′-deoxyCytidine,3′-[(2-cyanoethyl)-(N,N-diisopropyl)]-phosphoramidite | |
| FWC | 5-Br-dC-CE | 5′-Dimethoxytrityl-N-benzoyl-5-bromo-2′-deoxyCytidine,3′-[(2-cyanoethyl)-(N,N-diisopropyl)]-phosphoramidite | |
| FYC | 5-Carboxy-dC-CE | 5′-Dimethoxytrityl-N-benzoyl-5-ethylcarboxy-2′-deoxyCytidine,3′-[(2-cyanoethyl)-(N,N-diisopropyl)]-phosphoramidite | |
| FZC | 5-Hydroxymethyl-dC-CE | 5′-Dimethoxytrityl-N-benzoyl-5-cyanoethoxy-methyl-2′-deoxyCytidine,3′-[(2-cyanoethyl)-(N,N-diisopropyl)]-phosphoramidite | |
| FTC | 5-Me-dC-CE | 5′-Dimethoxytrityl-N-benzoyl-5-methyl-2′-deoxyCytidine,3′-[(2-cyanoethyl)-(N,N-diisopropyl)]-phosphoramidite |
G-based modification
T-based modification
| FRAGMENT ID | 2D Structure | Name (3D link) | Notes |
| DTT | Cytidine | Ideal module Name HARDCODED Charges from OL3/OL15 | |
| APT | alpha-putrescinylthymine | ||
| DHT | 5,6-dihydrothymine | ||
| M3T | 3-methylthymine | ||
| O4T | O(4)-methylthymine |